1. Intro to General Chemistry
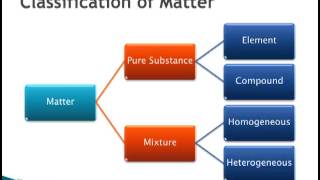
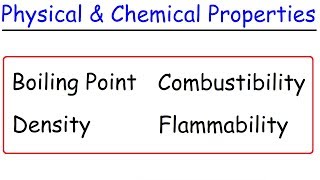
Physical Properties
Problem 20a
Textbook Question
Textbook Question(a) Read the following description of the element zinc and indicate which are physical properties and which are chemical properties. Zinc melts at 420 C. When zinc granules are added to dilute sulfuric acid, hydrogen is given off and the metal dissolves. Zinc has a hardness on the Mohs scale of 2.5 and a density of 7.13 g>cm3 at 25 C. It reacts slowly with oxygen gas at elevated temperatures to form zinc oxide, ZnO.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
852
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos