20. Electrochemistry
Electroplating
Problem 161
Textbook Question
Textbook QuestionExperimental solid-oxide fuel cells that use butane (C4H10) as the fuel have been reported recently. These cells contain composite metal/metal oxide electrodes and a solid metal oxide electrolyte. The cell half-reactions are
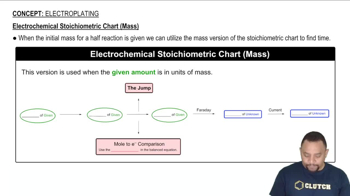
(c) How many grams of butane are required to produce a constant current of 10.5 A for 8.00 h? How many liters of gaseous butane at 20 °C and 815 mm Hg pressure are required?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
505
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 6 videos