13. Liquids, Solids & Intermolecular Forces
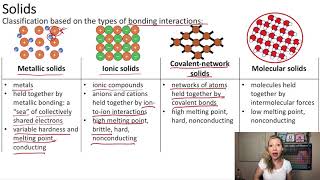
Atomic, Ionic, and Molecular Solids
Problem 56
Textbook Question
Textbook QuestionFor each of the following groups, which metal would you expect to have the highest melting point: (b) rubidium, molybdenum, or indium?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
782
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos