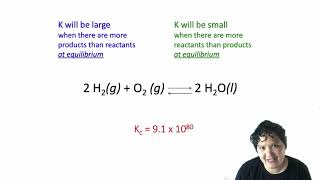16. Chemical Equilibrium
Reaction Quotient
Problem 81
Textbook Question
Textbook QuestionConsider the reaction: H2(g) + I2(g) ⇌ 2 HI(g) A reaction mixture at equilibrium at 175 K contains PH2 = 0.958 atm, PI2 = 0.877 atm, and PHI = 0.020 atm. A second reaction mixture, also at 175 K, contains PH2 = PI2 = 0.621 atm and PHI = 0.101 atm. Is the second reac- tion at equilibrium? If not, what will be the partial pressure of HI when the reaction reaches equilibrium at 175 K?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
2866
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos