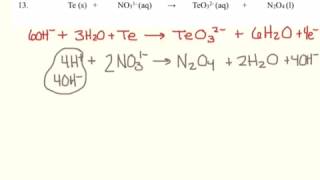
6. Chemical Quantities & Aqueous Reactions
Balancing Redox Reactions: Basic Solutions
Multiple Choice
Multiple ChoiceBalance the following redox reaction in a basic solution.
ClO2− (aq) → Cl- (aq) + ClO4− (aq)
A
3 ClO2− (aq) → 2 Cl− (aq) + ClO4− (aq)
B
2 ClO2− (aq) → Cl− (aq) + ClO4− (aq)
C
4 ClO2− (aq) → Cl− (aq) + 3 ClO4− (aq)
D
4 ClO2− (aq) → 2 Cl− (aq) + 2 ClO4− (aq)
1363
views
6
rank
1
comments
Related Videos
Related Practice
Showing 1 of 9 videos