3. Chemical Reactions
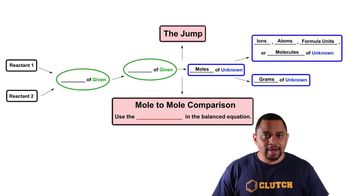
Limiting Reagent
Multiple Choice
Multiple ChoiceThe following reaction shows the mineral ilmenite, FeTiO3, being reacted with chlorine gas and sand in order to extract titanium (IV) chloride.
FeTiO3 (s) + 3 Cl2 (g) + 3 C (s) → 3 CO (g) + FeCl2 (s) + TiCl4 (g)
Assuming a 100% yield, how many grams of titanium (IV) chloride can be extracted when reacting 18.0 g ilmenite, 30.0 g Cl2 and 40.0 g C?
A
211 g
B
26.8 g
C
22.5 g
D
92.9 g
202
views
1
comments
Related Videos
Related Practice
Showing 1 of 11 videos