13. Liquids, Solids & Intermolecular Forces
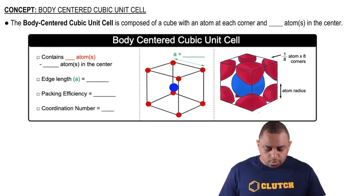
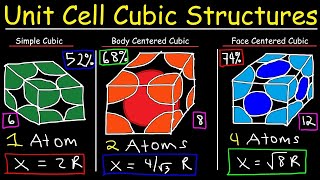
Body Centered Cubic Unit Cell
Multiple Choice
Multiple ChoiceVanadium has a body-centered cubic structure. If the atomic radius of vanadium is 134 pm, calculate the density of solid vanadium.
A
5.33 g/cm3
B
4.29 g/cm3
C
5.71 g/cm3
D
0.52 g/cm3
1182
views
2
rank
Related Videos
Related Practice
Showing 1 of 7 videos