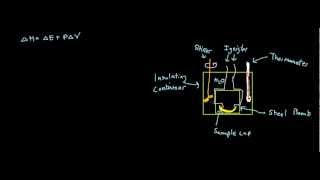
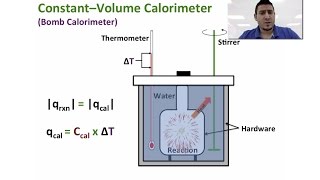
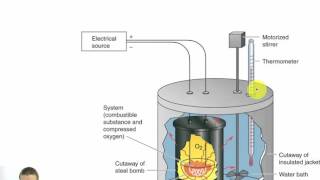
8. Thermochemistry
Constant-Volume Calorimetry
Multiple Choice
Multiple ChoiceWhen water evaporates from a beaker at 25℃, 44.0 kJ/mol of heat is required. How much heat is required when 10.0 g of water completely evaporates?
H2O (l) → H2O (g); ΔH = +44.0 kJ/mol
A
440 kJ
B
7920 kJ
C
24.4 kJ
D
79.3 kJ
E
Heat is released by the system (the water) in this instance.
406
views
Related Videos
Related Practice
Showing 1 of 10 videos