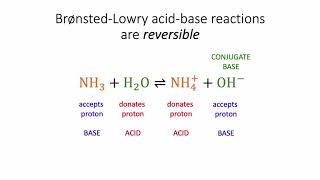17. Acid and Base Equilibrium
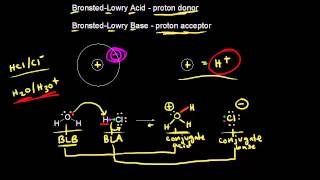
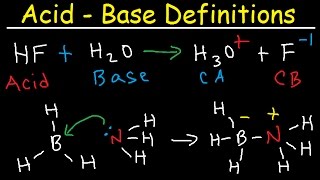
Bronsted-Lowry Acids and Bases
Problem 59
Textbook Question
Textbook QuestionWhich base in each of the following pairs has the stronger conjugate acid? See Table 16.1 to compare the relative strengths of conjugate acid-base pairs. (a) Cl- or CO32-
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
59sPlay a video:
371
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos