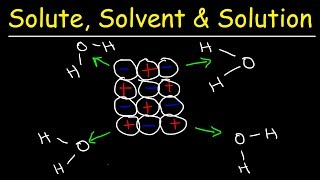
14. Solutions
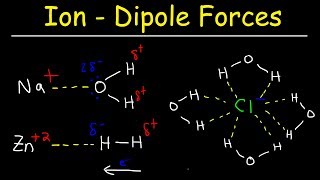
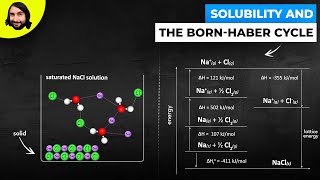
Solutions: Solubility and Intermolecular Forces
Problem 92
Textbook Question
Textbook QuestionA supersaturated solution of sucrose (C12H22O11) is made by dissolving sucrose in hot water and slowly letting the solution cool to room temperature. After a long time, the excess sucrose crystallizes out of the solution. Indicate whether each of the following statements is true or false: (b) After the excess sucrose has crystallized out, the system is now unstable and is not in equilibrium.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1353
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos