24. Transition Metals and Coordination Compounds
Strong-Field vs Weak-Field Ligands
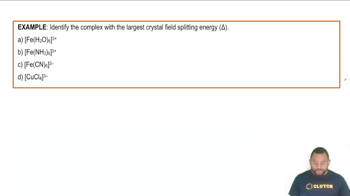
Problem 139c
Textbook Question
Textbook QuestionChromium forms three isomeric compounds A, B, and C with percent composition 19.52% Cr, 39.91% Cl, and 40.57% H2O. When a sample of each compound was dissolved in water and aqueous AgNO3 was added, a precipitate of AgCl formed immediately. A 0.225-g sample of compound A gave 0.363 g of AgCl, 0.263 g of B gave 0.283 g of AgCl, and 0.358 g of C gave 0.193 g of AgCl. One of the three compounds is violet, while the other two are green. In all three, chromium has coordination number 6. (c) Which of the three compounds is likely to be the violet one? Explain.
77
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos