2. Atoms & Elements
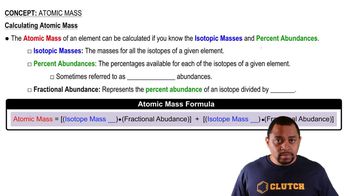
Atomic Mass
Open Question
Open QuestionBoron obtained from borax deposits in death valley consists of two isotopes. They are boron-10 and boron-11 with atomic masses of 10.013 amu and 11.009 amu, respectively. The atomic mass of boron is 10.81 amu (see periodic table). Which isotope of boron is more abundant, boron-10 or boron-11?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
191
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos