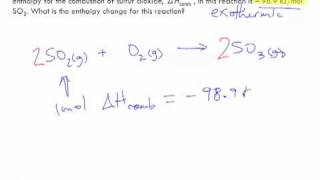8. Thermochemistry
Thermochemical Equations
Multiple Choice
Multiple ChoiceNitromethane (CH3NO2), sometimes used as a fuel for drag racing, burns according to the following reaction:
4 CH3NO2 (l) + 7 O2 (g) → 4 CO2 (g) + 6 H2O (g) + 4 NO2 (g) ∆Hº = – 2441.6 kJ
How much heat is released by burning 125.0 g of nitromethane (MW:61.044 g/mol)?
A
1.250 x 103 kJ
B
-1.250 x 103 kJ
C
2.440 x 103 kJ
D
-2.440 x 103 kJ
1793
views
2
rank
1
comments
Related Videos
Related Practice
Showing 1 of 10 videos