20. Electrochemistry
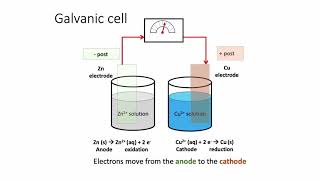
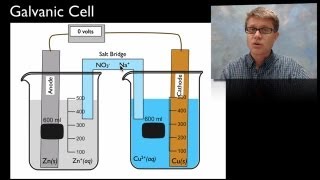
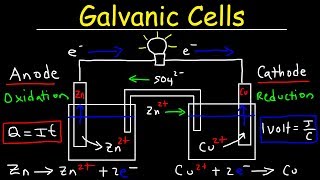
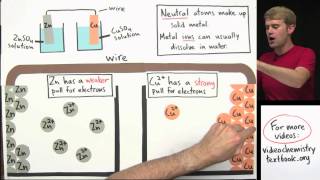
Galvanic Cell
Problem 4
Textbook Question
Textbook QuestionAssume that you want to construct a voltaic cell that uses the following half-reactions: A2+1aq2 + 2 e- ¡ A1s2 Ered ° = -0.10 V B2+1aq2 + 2 e- ¡ B1s2 E°red = -1.10 V You begin with the incomplete cell pictured here in which the electrodes are immersed in water.

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
651
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos