20. Electrochemistry
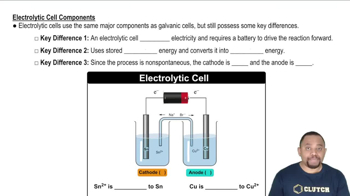
Electrolytic Cell
Problem 12
Textbook Question
Textbook QuestionMagnesium, the element, is produced commercially by electrolysis from a molten salt (the 'electrolyte') using a cell similar to the one shown here. (c) Recall that in an electrolytic cell the anode is given the + sign and the cathode is given the – sign, which is the opposite of what we see in batteries. What half-reaction occurs at the anode in this electrolytic cell?

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
820
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 6 videos