17. Acid and Base Equilibrium
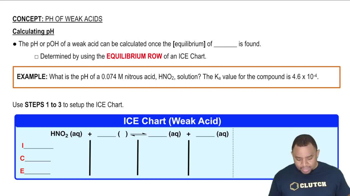
pH of Weak Acids
Problem 39b
Textbook Question
Textbook QuestionThe following pictures represent initial concentrations in solutions that contain a weak acid HA (pKa = 6.0) and its sodium salt NaA. (Na+ ions and solvent water molecules have been omitted for clarity.)

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
493
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 5 videos