24. Transition Metals and Coordination Compounds
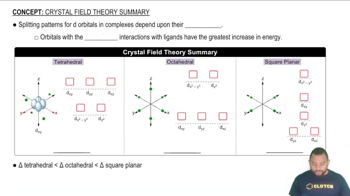
Crystal Field Theory Summary
Problem 21.131a
Textbook Question
Textbook QuestionNickel(II) complexes with the formula NiX2L2, where X− is Cl− or N-bonded NCS− and L is the monodentate triphenylphosphine ligand P(C6H5)3, can be square planar or tetrahedral.
(a) Draw crystal field energy-level diagrams for a square planar and a tetrahedral nickel(II) complex, and show the population of the orbitals.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
95
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos