7. Gases
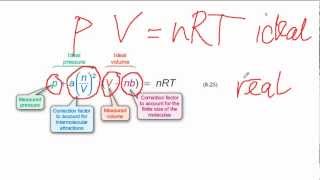
Van der Waals Equation
Problem 96
Textbook Question
Textbook QuestionTable 10.3 shows that the van der Waals b parameter has units of L/mol. This means that we can calculate the sizes of atoms or molecules from the b parameter. Refer back to the discussion in Section 7.3. Is the van der Waals radius we calculate from the b parameter of Table 10.3 more closely associated with the bonding or nonbonding atomic radius discussed there? Explain.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1133
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos