13. Liquids, Solids & Intermolecular Forces
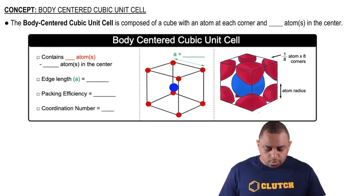
Body Centered Cubic Unit Cell
Problem 58b
Textbook Question
Textbook QuestionThe unit cell of a compound containing Co and O has a unit cell shown below. The Co atoms are on the corners, and the O atoms are completely within the unit cell. What is the empirical formula of this compound? What is the oxidation state of the metal?

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
663
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 7 videos