18. Aqueous Equilibrium
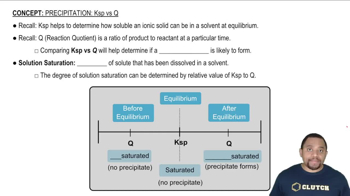
Precipitation: Ksp vs Q
Problem 139
Textbook Question
Textbook QuestionIn quantitative analysis, Ag+, Hg2+, and Pb2+ are seperated from other cations by the addition of HCl. Calculate the concentration of Cl-ions required to just begin the precipitation of (a) AgCl, (b) Hg2Cl2, (c) PbCl2 in a solution hav-ing metal-ion concentrations of 0.030 M. What fraction of the Pb2+ remains in solution when the Ag+ just begins to precipitate?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
1472
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos