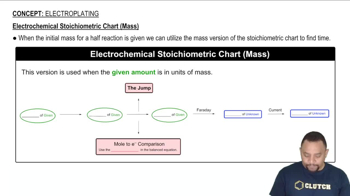
20. Electrochemistry
Electroplating
Problem 127b
Textbook Question
Textbook QuestionA current of 11.3 A is applied to 1.25 L of a solution of 0.552 M HBr converting some of the H+ to H2(g), which bubles out of solution. What is the pH of the solution after 73 minutes?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
792
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 6 videos