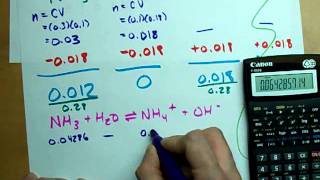18. Aqueous Equilibrium
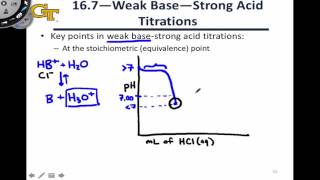
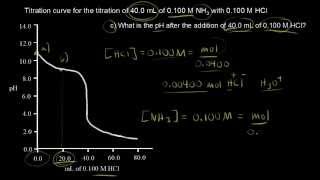
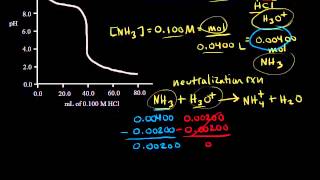
Titrations: Weak Base-Strong Acid
Multiple Choice
Multiple ChoiceIn order to create a buffer 7.321 g of potassium lactate is mix with 550.0 mL of 0.328 M lactic acid, HC3H5O3. What is the pH of the buffer solution after the addition of 300.0 mL of 0.100 M hydrobromic acid, HBr? The Ka of HC3H5O3 is 1.4 × 10−4.
A
3.16
B
2.96
C
4.74
D
4.35
194
views
1
rank
Related Videos
Related Practice
Showing 1 of 12 videos