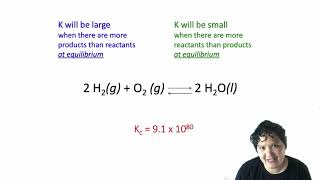16. Chemical Equilibrium
Reaction Quotient
Multiple Choice
Multiple ChoiceFor the reaction: 2 CO2 (g) ⇌ 2 CO (g) + 2 O2 (g), the equilibrium constant is 3.12 × 10−4 at 400 K, while the reaction quotient is 4.18 × 10−4. If initially we have 0.20 atm CO2, 0.30 atm CO and 0.15 atm O2, which of the following statements is not true?
a) The pressure of CO2 will be greater than 0.20 atm.
b) The pressure of CO will be less than 0.30 atm.
c) The pressure of O2 will be greater than 0.15 atm.
d) The pressure of O2 will be less than 0.15 atm.
e) The reaction will favor reactants.
A
The pressure of CO2 will be greater than 0.20 atm.
B
The pressure of CO will be less than 0.30 atm.
C
The pressure of O2 will be greater than 0.15 atm.
D
The pressure of O2 will be less than 0.15 atm.
E
The reaction will favor reactants.
223
views
1
rank
Related Videos
Related Practice
Showing 1 of 10 videos