18. Aqueous Equilibrium
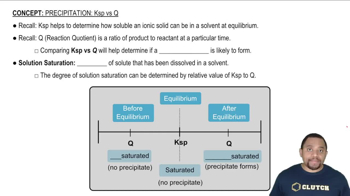
Precipitation: Ksp vs Q
Problem 74a
Textbook Question
Textbook QuestionA solution of Na2SO4 is added dropwise to a solution that is 0.010 M in Ba2 +1aq2 and 0.010 M in Sr2 +1aq2. (a) What concentration of SO42 - is necessary to begin precipitation? (Neglect volume changes. BaSO4: Ksp = 1.1 * 10-10; SrSO4: Ksp = 3.2 * 10-7.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
1404
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 2 videos