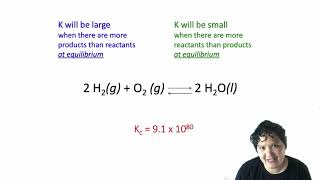16. Chemical Equilibrium
Reaction Quotient
Problem 49
Textbook Question
Textbook QuestionSilver sulfate dissolves in water according to the reaction: Ag2SO4(s) ⇌ 2Ag+(aq) + SO42-(aq) Kc = 1.1 * 10-5 at298K A 1.5-L solution contains 6.55 g of dissolved silver sulfate. If addi- tional solid silver sulfate is added to the solution, will it dissolve?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
5062
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos