19. Chemical Thermodynamics
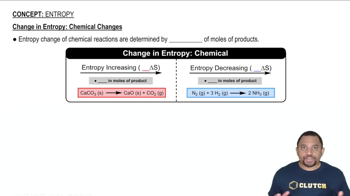
Entropy
Problem 3
Textbook Question
Textbook QuestionIf energy can flow in and out of the system to maintain a constant temperature during the process, what can you say about the entropy change of the surroundings as a result of this process?

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
378
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos