2. Atoms & Elements
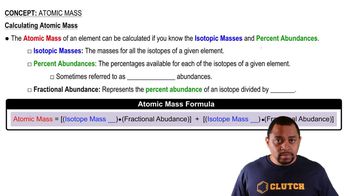
Atomic Mass
Problem 98b
Textbook Question
Textbook QuestionThere are two different isotopes of bromine atoms. Under normal conditions, elemental bromine consists of Br2 molecules, and the mass of a Br2 molecule is the sum of the masses of the two atoms in the molecule. The mass spectrum of Br2 consists of three peaks: Mass (u) Relative Size 157.836 0.2569 159.834 0.4999 161.832 0.2431 (b) What is the mass of each isotope?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
2492
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos