14. Solutions
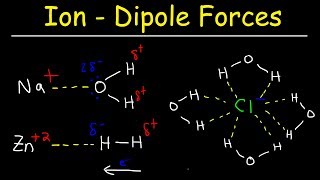
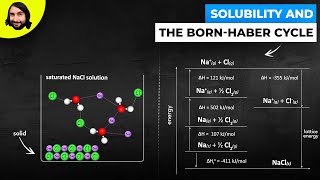
Solutions: Solubility and Intermolecular Forces
Problem 31
Textbook Question
Textbook QuestionWhich molecule would you expect to be more soluble in water: CH3CH2CH2OH or HOCH2CH2CH2OH?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
3963
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 16 videos