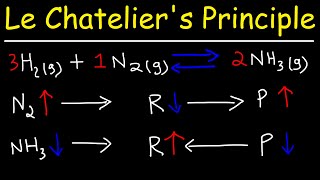
16. Chemical Equilibrium
Le Chatelier's Principle
Problem 132
Textbook Question
Textbook QuestionA platinum catalyst is used in automobile catalytic convert- ers to hasten the oxidation of carbon monoxide: 2 CO1g2 + O 1g2 ∆Pt 2 CO 1g2 ΔH° = - 566 kJ Suppose that you have a reaction vessel containing an equilibrium mixture of CO1g2, O21g2, and CO21g2. Under the following conditions, will the amount of CO increase, decrease, or remain the same? (e) The pressure is increased by adding O2 gas.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
489
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 13 videos