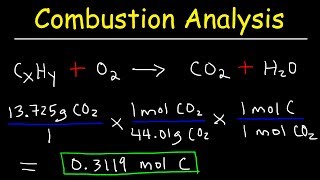
3. Chemical Reactions
Molecular Formula
Multiple Choice
Multiple ChoiceElemental analysis of a pure compound indicated that the compound had 72.2% C, 8.50% H and the remainder as O. If 0.250 moles of the compound weighs 41.55 g, what is the molecular formula of the compound?
A
C5H7O
B
C10H14O2
C
C6H8O2
D
C12H16O4
796
views
11
rank
1
comments
Related Videos
Related Practice
Showing 1 of 8 videos