16. Chemical Equilibrium
ICE Charts
Problem 144
Textbook Question
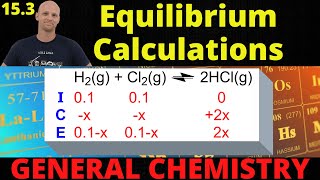
Textbook QuestionVinegar contains acetic acid, a weak acid that is partially dissociated in aqueous solution: CH3CO2H1aq2 ∆ H+ 1aq2 + CH3CO-1aq2 (b) What is the value of Kc if the extent of dissociation in 1.0 M CH3CO2H is 0.42%?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
631
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos