8. Thermochemistry
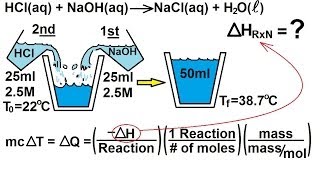
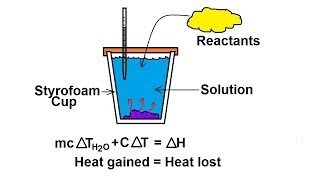
Constant-Pressure Calorimetry
Problem 88
Textbook Question
Textbook QuestionWhen 1.50 g of magnesium metal is allowed to react with 200 mL of 6.00 M aqueous HCl, the temperature rises from 25.0 °C to 42.9 °C. Calculate ΔH in kilojoules for the reaction, assumign that the heat capacity of the calorimeter is 776 J/°C, that the specific heat of the final soltuion is the same as that of water [4.18 J(g·°C)] and that the density of the solution is 1.00 g/mL
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
8mPlay a video:
755
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos