20. Electrochemistry
Galvanic Cell
Problem 79
Textbook Question
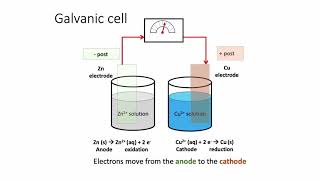
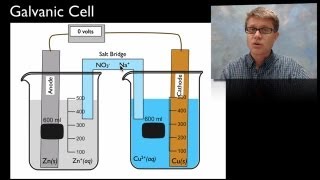
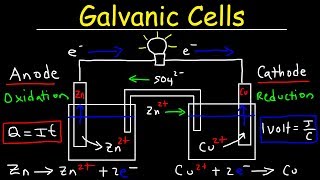
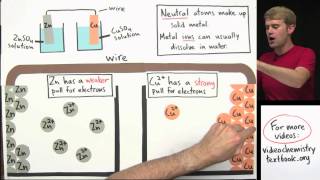
Textbook QuestionMake a sketch of a concentration cell employing two Zn/Zn2+ half-cells. The concentration of Zn2+ in one of the half-cells is 2.0 M and the concentration in the other half-cell is 1.0x10^-3 M. Label the anode and the cathode and indicate the half-reaction occuring at each electrode. Also indicate the direction of electron flow.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
975
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 15 videos