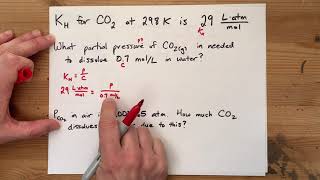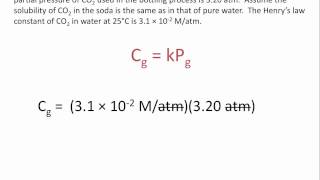14. Solutions
Henry's Law Calculations
Multiple Choice
Multiple ChoiceThe atmospheric pressure in a lab is calculated as 1.3 atm. If oxygen gas contributes 62% of this atmospheric pressure, determine its mass (in g) dissolved at room temperature in 25 L of water. The Henry's Law Constant for oxygen in water at this temperature is 5.3 × 10–5 M/atm.
A
1.4 × 10–3 g
B
6.9 × 10–5 g
C
0.055 g
D
0.034 g
1369
views
6
rank
Related Videos
Related Practice
Showing 1 of 10 videos