8. Thermochemistry
Enthalpy of Formation
Multiple Choice
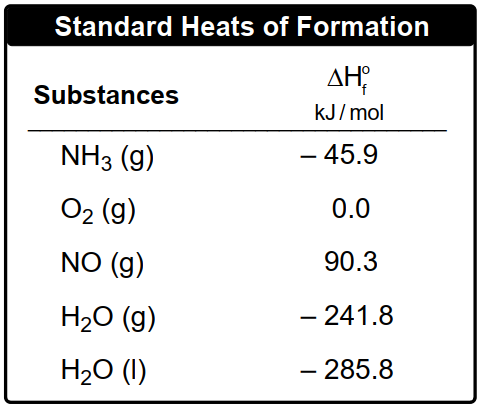
Multiple ChoiceThe oxidation of ammonia is illustrated by the following equation:
4 NH3 (g) + 5 O2 (g) → 4 NO (g) + 6 H2O (g)
Calculate the enthalpy of reaction, ΔHRxn, based on the given standard heats of formation.
A
-906 kJ
B
-1273.2 kJ
C
1089.6 kJ
D
-183.6 kJ
2169
views
Related Videos
Related Practice
Showing 1 of 6 videos