16. Chemical Equilibrium
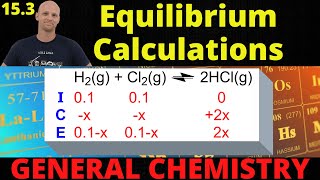
ICE Charts
Multiple Choice
Multiple ChoiceAt a given temperature the gas phase reaction:N2 (g) + O2 (g) ⇌ 2 NO (g) has an equilibrium constant of 4.00 x 10 -15. What will be the concentration of NO at equilibrium if 2.00 moles of nitrogen and 6.00 moles oxygen are allowed to come to equilibrium in a 2.0 L flask.
A
1.10 x 10-7 M
B
5.48 x 10 -7 M
C
5.48 x 10 -8 M
D
1.10 x 10-8 M
1704
views
1
rank
17
comments
Related Videos
Related Practice
Showing 1 of 12 videos