13. Liquids, Solids & Intermolecular Forces
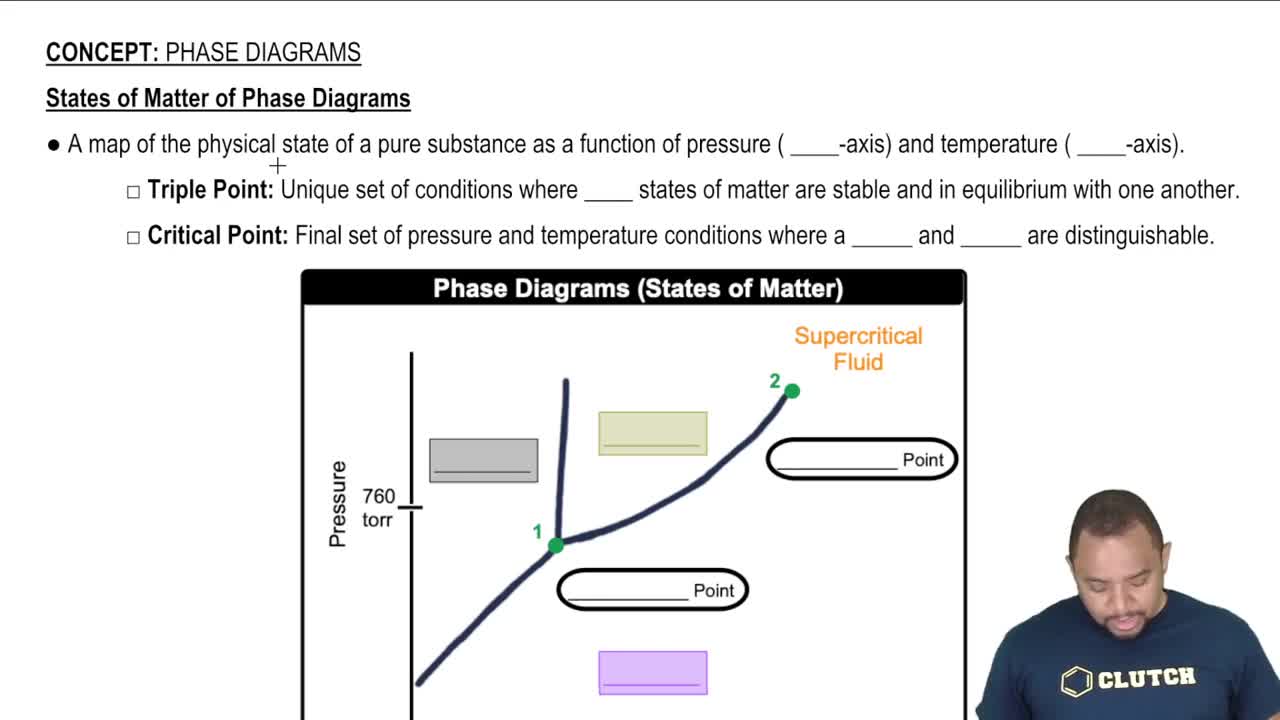
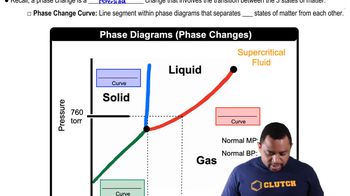
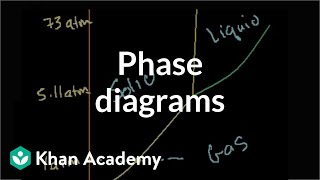
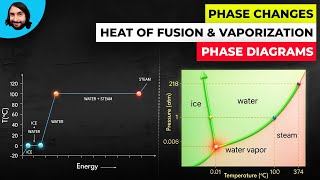
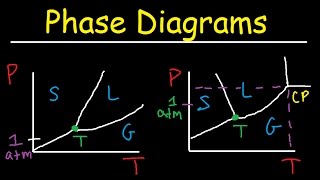
Phase Diagrams
Problem 64
Textbook Question
Textbook QuestionAt 25 °C gallium is a solid with a density of 5.91 g/cm3. Its melting point, 29.8 °C, is low enough that you can melt it by holding it in your hand. The density of liquid gallium just above the melting point is 6.1 g/cm3. Based on this information, what unusual feature would you expect to find in the phase diagram of gallium?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
1mPlay a video:
1188
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos