12. Molecular Shapes & Valence Bond Theory
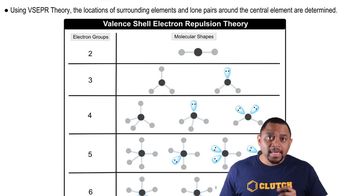
Valence Shell Electron Pair Repulsion Theory
Multiple Choice
Multiple ChoiceWhich of the following statements is false about VSEPR theory?
A
Each of the following counts as a single electron group: a lone pair, a single bond, a double bond, a triple bond, and a single electron.
B
The geometry of the electron groups is determined by minimizing their repulsions.
C
The number of electron groups can be determined from the Lewis structure of the molecule.
D
Bond angles can vary from the idealized angles because lone pairs of electrons take up less space than bonding groups.
E
The geometry of a molecule is determined by the number of electron groups on the central atom.
307
views
Related Videos
Related Practice
Showing 1 of 11 videos