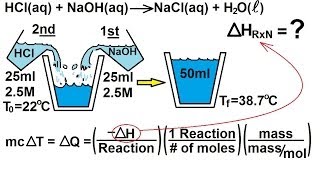
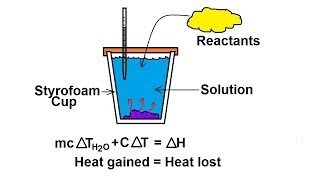
8. Thermochemistry
Constant-Pressure Calorimetry
Problem 153
Textbook Question
Textbook Question(c) Assume that a chunk of potassium weighing 7.55 g is dropped into 400.0 g of water at 25.0 °C. What is the final temperature of the water if all the heat released is used to warm the water?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
382
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos