20. Electrochemistry
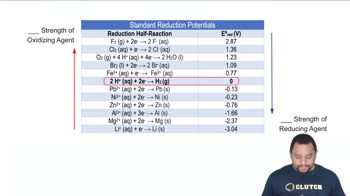
Standard Reduction Potentials
Multiple Choice
Multiple ChoiceRank the given metal ions in order of increasing strength as an oxidizing agent.
Pb2+ (-0.13 V), Mn2+ (-1.18 V), Cu2+ (+0.16 V), Co3+ (+1.82 V), Fe3+ (+0.77 V)
A
Pb2+ < Cu2+ < Mn2+ < Co3+ < Fe3+
B
Co3+ < Fe3+ < Cu2+ < Pb2+ < Mn2+
C
Mn2+ < Pb2+ < Cu2+ < Fe3+ < Co3+
D
Mn2+ < Pb2+ < Cu2+ < Co3+ < Fe3+
247
views
Related Videos
Related Practice
Showing 1 of 10 videos