13. Liquids, Solids & Intermolecular Forces
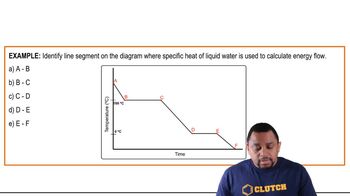
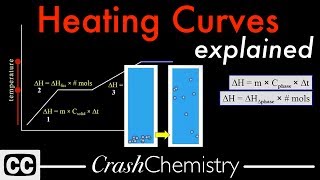
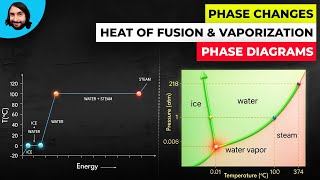
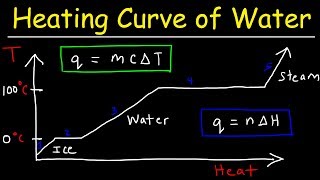
Heating and Cooling Curves
Multiple Choice
Multiple ChoiceHow much energy (kJ) is required to convert a 76.4 g acetone (MM = 58.08 g/mol) as a liquid at -30°C to a solid at -115.0°C?
A
-11.406 kJ
B
-39.820 kJ
C
-22.811 kJ
D
-82.592 kJ
1054
views
3
comments
Related Videos
Related Practice
Showing 1 of 10 videos