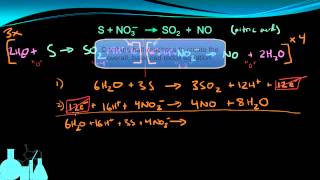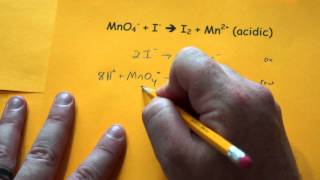6. Chemical Quantities & Aqueous Reactions
Balancing Redox Reactions: Acidic Solutions
Problem 155a
Textbook Question
Textbook QuestionSodium nitrite, NaNO2, is frequently added to processed meats as a preservative. The amount of nitrite ion in a sample can be determined by acidifying to form nitrous acid (HNO2), letting the nitrous acid react with an excess of iodide ion, and then titrating the I3 - ion that results with thiosulfate solution in the presence of a starch indicator. The unbalanced equations are (1) (2) (b) When a nitrite-containing sample with a mass of 2.935 g was analyzed, 18.77 mL of 0.1500 M Na2S2O3 solution was needed for the reaction. What is the mass percent of NO2- ion in the sample?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
23mPlay a video:
586
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos