19. Chemical Thermodynamics
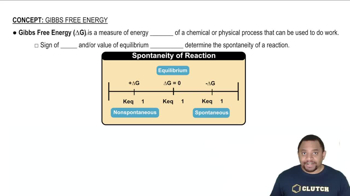
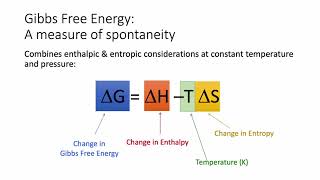
Gibbs Free Energy
Problem 7b
Textbook Question
Textbook QuestionIdentify the true statement about a spontaneous process. (a) A reaction that is nonspontaneous in the forward direction is spontaneous in the reverse direction. (b) Adding a catalyst will cause a nonspontaneous reaction to become spontaneous. (c) In a spontaneous process, the entropy of the system always decreases. (d) An endothermic reaction is always spontaneous.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
577
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos