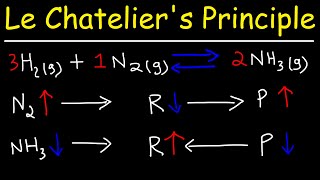
16. Chemical Equilibrium
Le Chatelier's Principle
Multiple Choice
Multiple ChoiceWhich direction will the following reaction (in a 10.0 L flask) proceed if a catalyst is added to the system?
CaCO3 (s) ⇌ CaO (s) + CO2 (g) Kp = 3.2 x 10-28
A
To the right.
B
To the left.
C
The equilibrium position will not change but the rate will increase.
D
The equilibrium position will not change but the concentrations of everything will increase.
796
views
3
rank
1
comments
Related Videos
Related Practice
Showing 1 of 13 videos