19. Chemical Thermodynamics
Entropy
Multiple Choice
Multiple ChoiceWhich reaction is most likely to have a positive ∆S of reaction?
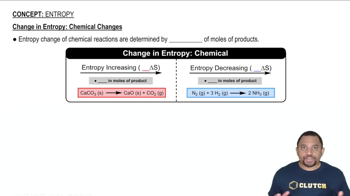
a) SiO2 (s) + 3 C (s) → SiC (s) + 2 CO (g)
b) 6 CO2 (g) + 6 H2O (g) → C6H12O6 (s) + 6 O2 (g)
c) CO (g) + Cl2 (g) → COCl2 (g)
d) 3 NO2 (g) + H2O (l) → 2 HNO3 (l) + NO (g)
A
SiO2 (s) + 3 C (s) → SiC (s) + 2 CO (g)
B
6 CO2 (g) + 6 H2O (g) → C6H12O6 (s) + 6 O2 (g)
C
CO (g) + Cl2 (g) → COCl2 (g)
D
3 NO2 (g) + H2O (l) → 2 HNO3 (l) + NO (g)
285
views
2
rank
1
comments
Related Videos
Related Practice
Showing 1 of 16 videos