16. Chemical Equilibrium
ICE Charts
Problem 111
Textbook Question
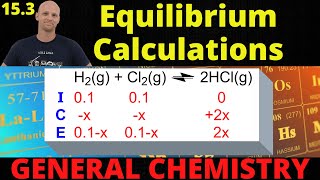
Textbook QuestionThe equilibrium constant Kc for the reaction N21g2 + 3 H21g2 ∆ 2 NH31g2 is 4.20 at 600 K. When a quantity of gaseous NH3 was placed in a 1.00-L reaction vessel at 600 K and the reaction was allowed to reach equilibrium, the vessel was found to contain 0.200 mol of N2. How many moles of NH3 were placed in the vessel?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
3mPlay a video:
427
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos