13. Liquids, Solids & Intermolecular Forces
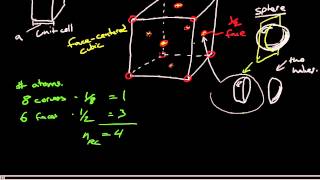
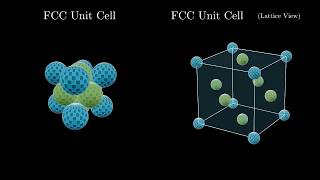
Face Centered Cubic Unit Cell
Multiple Choice
Multiple ChoiceAn element crystallizes in a face-centered cubic lattice and has a density of 18.44 g/cm3. The edge of its unit cell is 1.05×10-8 cm. Calculate the atomic mass for the element.
A
3.21 g•mol-1
B
15.5 g•mol-1
C
60.8 g•mol-1
D
151 g•mol-1
754
views
Related Videos
Related Practice
Showing 1 of 7 videos