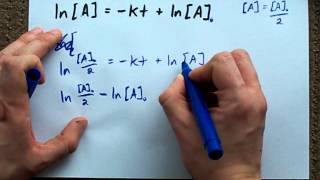21. Nuclear Chemistry
Rate of Radioactive Decay
Problem 55
Textbook Question
Textbook QuestionIodine-131 is a convenient radioisotope to monitor thyroid activity in humans. It is a beta emitter with a half-life of 8.02 days. The thyroid is the only gland in the body that uses iodine. A person undergoing a test of thyroid activity drinks a solution of NaI, in which only a small fraction of the iodide is radioactive. (c) A normal thyroid will take up about 12% of the ingested iodide in a few hours. How long will it take for the radioactive iodide taken up and held by the thyroid to decay to 0.01% of the original amount?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
883
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos