10. Periodic Properties of the Elements
Periodic Trend: Electron Affinity
Problem 98
Textbook Question
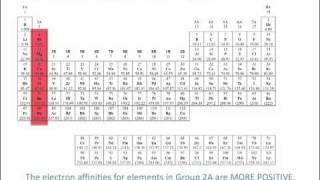
Textbook QuestionThe electron affinities, in kJ>mol, for the group 11 and group 12 metals are as follows: Cu -119 Zn 7 0 Ag -126 Cd 7 0 Au -223 Hg 7 0 (b) Why do the electron affinities of the group 11 elements become more negative as we move down the group? [Hint: Examine the trends in the electron affinities of other groups as we proceed down the periodic table.]

 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
2mPlay a video:
1331
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 14 videos