16. Chemical Equilibrium
ICE Charts
Problem 156
Textbook Question
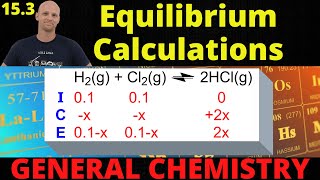
Textbook QuestionA 79.2 g chunk of dry ice (solid CO2) and 30.0 g of graphite (carbon) were placed in an empty 5.00-L container, and the mixture was heated to achieve equilibrium. The reaction is CO 1g2 + C s ∆ 2 CO g (b) What is the value of Kp at 1100 K if the gas density at 1100 K is 16.9 g/L?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
8mPlay a video:
789
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 12 videos